Thirty years ago, the British Retail Consortium established global standard to respond to the increased audit burden created by the proliferation in retailer manufacturing standards introduced in the 1990’s. It is fair to say that a lot has changed over the intervening years but third-party certificated audit schemes have become established as a key part of product safety management programmes throughout the entire global supply chain. This article takes a look back at the last 30 years and how product safety management has evolved to respond to the established and emerging hazards in an ever-changing product supply system.
Cultural and product safety events
Cast your mind back to 1996 and you might remember singing the songs of the Spice Girls or dancing to the Macarena. Sports fans will have been celebrating Michael Johnson winning the 200m and 400m at the Atlanta Olympics and Damon Hill winning the F1 Championship. The Nokia 1011 remained one of the most popular mobile phones, 10 years before the first iPhone entered the market. The scientists among you may have been marvelling at the cloning of Dolly the Sheep at the Roslin Institute, the sequencing of the first eukaryotic cell, the yeast Saccharomyces cerevisiae or the sale of the first genetically modified food product, tomato paste in the UK. Perhaps the most sobering statistic was the global population in 1996 being approximately 5.8bn, 2.4bn lower than the estimate for 2025 (8.2bn).
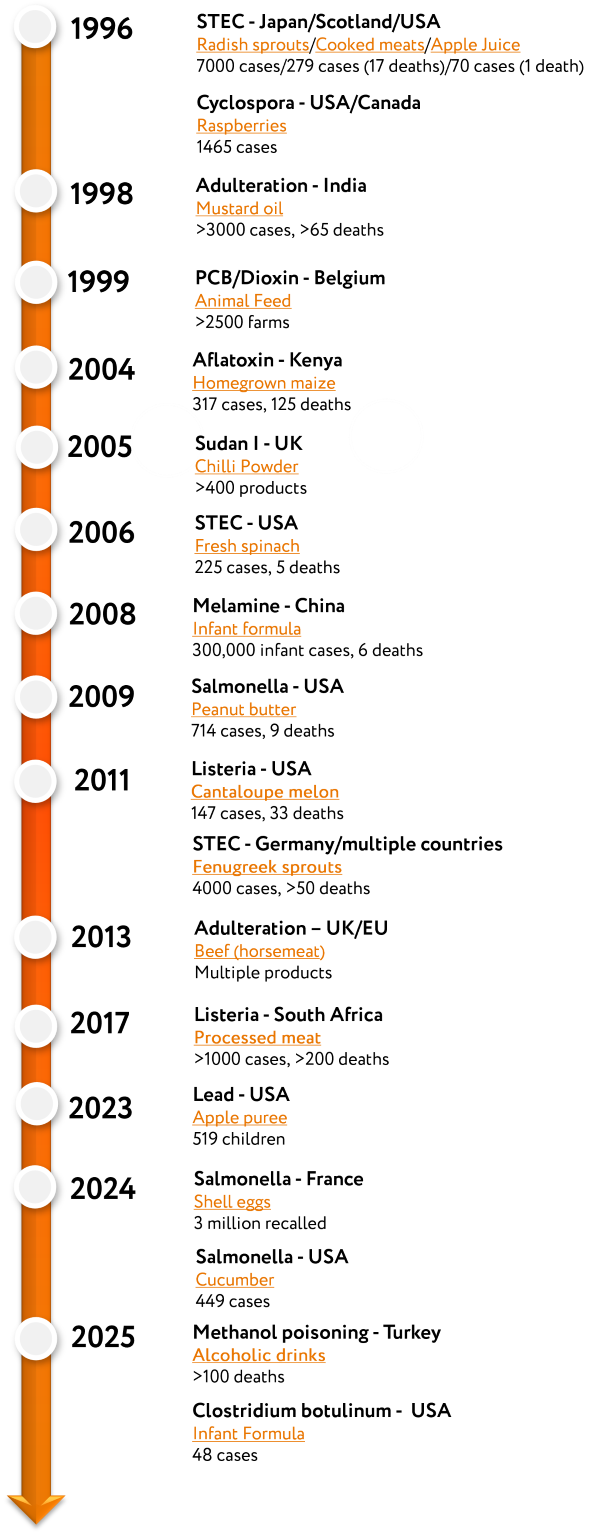
Also in 1996, the UK announced a probable link between Bovine Spongiform Encephalopathy (BSE) and Creutzfeldt-Jakob Disease (vCJD) which led to a global ban on British beef. Shiga toxin-producing E. coli (STEC) caused three devastating global outbreaks linked to white radish sprouts in Japan, unpasteurised apple juice in the USA and cooked meats in the UK. In addition to STEC, the major burden of foodborne infectious disease included Salmonella spp., especially S. Enteritidis and S. Typhimurium, Campylobacter spp., Listeria monocytogenes and norovirus. Sadly, all of these remain the most significant causes of infectious intestinal disease in most industrialised countries. Some of the highest profile incidents of microbiological contamination since 1996 are shown in Figure 1.
|
Figure 1: Timeline of food safety events (see PDF version to access links) |
Chemical hazards in foods have been around for many centuries, often driven by adulteration and authenticity, and in 1996 a report outlined lead intoxication due to foods containing ground paprika contaminated with lead tetroxide (red lead).
Over the years chemical adulteration has included mustard oil contaminated with white oil that caused thousands of illnesses in India (1998), contamination of chilli powder with Sudan I in the UK (around 2003) and the adulteration of milk powder with melamine in China that caused hundreds of thousands of cases (2008). Chemical contamination with natural toxicants such as mycotoxins frequently feature as drivers of recalls but they can cause acute disease outbreaks when present at high levels as seen in Kenya in 2004 with homegrown maize.
Consumer products have not been immune to product safety issues over the years with incidents such as choking, fire, electrocution and heavy metal contamination being regular drivers of recalls. Developments in consumer products in the last 30 years especially with technological advancements and the widespread access to low cost, mass manufactured products have presented risk management challenges for both formulated and fabricated products. Incidents from traditional risks such as heavy metals like lead in toys, jewellery and clothing, appliance fires from white goods and microbiological contamination of face wipes demonstrate the continued issues faced by this sector. Fires associated with batteries in e-cigarettes, e-bikes and scooters plus internal injuries to children from high powered magnets in toys are recent examples that show how important it is for standards to evolve and a driver for the Global Standard Consumer Products to be updated in 2026.
Regulatory developments since 2006
Legislative requirements for foods and consumer products have focussed heavily on measures to enhance the control of product safety hazards. 1996 saw the introduction of new food labelling regulations in the UK requiring accurate labels to prevent fraudulent misrepresentation, such as incorrect ingredient listing, falsified origin, or misleading product names. Allergens, although recognised as a cause of a number of high-profile deaths for many years, were not subject to mandatory labelling at that time. Instead they were managed through voluntary labelling initiatives until the early 2000’s when countries (including the EU and the USA) introduced legislative requirements to name allergens and all ingredients on labels for food.
The early 2000’s saw harmonised regulations established in the EU for contaminants in foods including heavy metals, mycotoxins and dioxins. Additional hazards have been progressively added, often with decreased legislative limits as their effects on human health have become better understood.
In relation to microbiological hazards, 1996 was notable in the United States when the United States Department of Agriculture (USDA) introduced the final rule for Pathogen Reduction: Hazard Analysis and Critical Control Point (HACCP) Systems. This followed the major STEC outbreak associated with burgers from a quick service restaurant in the early 1990’s and mandated a hazard analysis and critical control point approach for all meat and poultry plants. The focus turned to prevention rather than just inspection.
The BSE crisis in the UK, the STEC outbreak in meat products and other high profile food incidents including Salmonella in eggs and listeriosis from Belgian pâté led to the introduction in the UK of the Food Safety Act (1990). The Act made food business operators directly responsible for ensuring product was safe, authentic and described accurately. The UK Food Standards Agency was formed in 2000 as a non-ministerial body to protect public health and restore consumer trust in the food system.
The EU followed with the introduction of Regulation EC 178/2002, laying down the general principles on food law that also made food business operators legally responsible for food safety and introducing strict traceability requirements. At the same time the European Food Safety Authority (EFSA) was formed. The EU made HACCP-based procedures mandatory in 2004 with the EU Hygiene Package (Regulation EC 852/2004 and 853/2004) and strengthened food labelling in the Food Information to Consumers Regulations (1169/2011). Regulation EC 2073/2005 set limits for a variety of microbial pathogens in foods as well as process hygiene criteria for a variety of primary agricultural products including meat, poultry, egg, dairy, fruit and vegetables.
In 2011, the US Food and Drug Administration (USFDA) signed into law the Food Safety Modernization Act (FSMA) to shift the focus from responding to contamination to preventing it. Key areas included preventive controls for human/animal food, produce safety standards and enhanced traceability.
Legislation for consumer products, applicable at both a product-specific level and across broader categories has evolved significantly since 1996. For example, the EU for cosmetics directive of 1976 was replaced in 2009 with Regulation 1223/2009 to recognise the potential risk associated with nanoparticles and introduce traceability requirements.
Developments reflect the modern challenges associated with digitalization and online marketplaces. The Toy Safety Directive (2009/48/EC) was introduced in 2009 to establish minimum safety standards regarding physical, chemical, and flammability risk. It required manufacturers to perform conformity assessments, compile technical documentation and affix a CE mark before sale. The Directive was updated in 2025 with tighter chemical limits, cybersecurity rules for connected/AI toys, strengthened warnings, online marketplace obligations and the use of digital product passports (Regulation 2025/2509).
Legislation covering a broader group of consumer products, transitioned from the 1992 General Product Safety Directive (Directive 92/59/EEC) to the 2001 Directive (2001/95/EC). This was replaced in 2024 with the General Product Safety Regulation (2023/988) which reflected the growth in digital sales and online markets.
Evolution of product safety assurance
It is fair to say that the product safety landscape has significantly evolved over the last 30 years with an ever-increasing range of hazards, risks and tighter regulatory controls reflecting a greater understanding of potential risks to health. The industry has responded to these challenges by developing more sophisticated and holistic approaches to product safety management with hazard analysis and risk assessment at the core.
Historical precedence and quality control
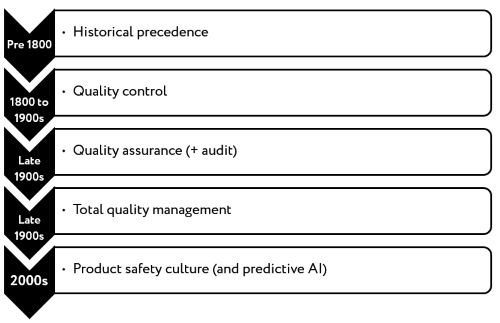
If you go back centuries, when hazards were poorly understood, controls were established based on ‘cause and effect’ creating a reliance on historical precedence. The development of analytical chemistry and the discovery of the microscope in the 17th and 18th centuries were the ‘technologies’ that heralded the first age of safety management based on analysis. Up to the late 1900’s, there was a significant reliance on product testing to ‘ensure’ the safety of product, the so-called ‘quality control’ approach to safety management. The assumption was that safety could be established by taking samples of the finished product, testing for the known hazards and establishing that the product was fit for sale. It became widely recognised that although testing could provide some indication of conformance, it could not be relied upon as a control in its own right.
|
Figure 2: Development of product safety management |
Quality assurance and product safety assurance
The advent of systems-based assurance or ‘quality/product safety assurance’ emerged in the late 1990’s. Product safety was achieved through the systematic identification of hazards, the understanding of controls necessary to eliminate or reduce the hazard to an acceptable level and the implementation of control measures in the manufacturing process. These controls, if consistently applied, would always deliver a safe product. This, of course, is the fundamental principle enshrined in HACCP and is universally recognised as the best approach to ensuring the safety of a product.
Testing or analysis still plays an important role, usually as part of verification that the HACCP plan delivers a product conforming to the standard. It also validates the efficacy of processes and product by, for example, establishing the conditions necessary for the destruction of micro-organisms or the microbiological stability of products like cosmetics.
Total quality management
Quality assurance paved a significant way forward to a more structured approach to product safety management but it did not fully overcome one of the most challenging aspects - driving an organisation-wide approach.
Total quality management (TQM) is a tool adopted in recent decades by industries that recognised the need to harness all personnel in the delivery of continuously improving operational practices with the common goal of improving product quality. Key elements of a TQM approach included the use of fact-based evidence, key performance indicators and continuous high-level communication to ensure full engagement of colleagues in the business. TQM has been adopted in many sectors and it both benefits and suffers from a very broad focus across all operations.
Product safety culture
Safety culture has its origins in all of the preceding approaches to product safety management. At its core is the recognition that the values in an organisation are pivotal to instilling a way of working that strives for the best outcome - product safety for the consumer. Product safety culture has been endorsed by many organisations including GFSI who have published an excellent position paper on food safety culture. Culture has been incorporated into GFSI-recognised product safety management certification programmes, including BRCGS.
The rise of the third-party certificated audit
Systems-based assurance is the foundation of product safety management systems that emerged in the 1990s and became embedded in standards developed by businesses for the safe production of their own products (first party) or their suppliers (second party). As these proliferated, businesses in the supply chain became burdened with product safety standards imposed by business customers, often with differing and sometimes conflicting requirements. In addition to the standard, auditing became a key mechanism to verify that the product safety management system was effective.
Initially, businesses conducted their own audits to their own standard to verify that their supply base had implemented requirements effectively and that hazards were under control. Second party audits proliferated throughout the supply chain.
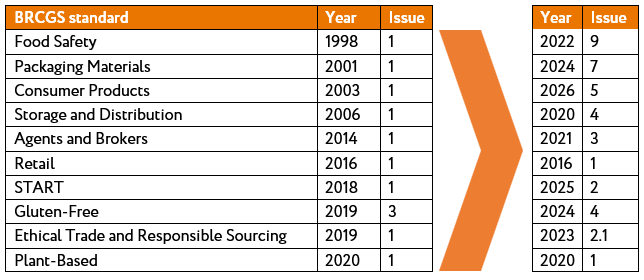
Third-party product safety management standards emerged to harmonise common product safety requirements across sectors in the supply chain. The first version of what ultimately became the BRCGS Global Standard Food Safety was published in 1998. Audits to demonstrate conformance began to predominate across the supply chain as retailers and manufacturers specified the third-party standards as a minimum requirement for supply to them.
Further third-party product safety management programmes began to emerge in the late 1990s across the world, particularly for food safety, and this provoked the need to establish a common standard for third-party programmes. The Global Food Safety Initiative (GFSI) was created to harmonise core requirements and provide a benchmark against which food safety programmes could be assessed. In 2000, the Global Standard for Food Safety became the first GFSI-recognised food safety management programme.
More than food
Third-party product safety management standards and programmes have been progressively developed for other sectors over the course of the last 30 years, with BRCGS pioneering many of these developments. In 2019, BRCGS separated from the BRC to develop the focus on global assurance issues. Issue 1 of the Global Standard Packaging Materials was published in 2001, soon to be followed by the Global Standard Consumer Products in 2003 and the Global Standard Storage and Distribution in 2006. Further BRCGS standards were published for agents and brokers in 2014, for smaller business looking for an entry level food safety management programme (START) in 2018 and for plant-based foods in 2020.
Product safety management standards and programmes have continuously improved over the years, to respond to emerging hazards and risks in the supply chain and the need for enhanced control. Certification programmes have been published by other organisations over the last 30 years and third-party product safety management certification is now established as an important component of product safety management programmes in businesses throughout the world.
|
Figure 3: Evolution of BRCGS global standards |
The future
Product safety management has evolved over the last 30 years to respond to the challenges inherent in the sourcing of ingredients, components and products from a global supply chain. The highly digitised systems operating in the majority of the supply chain makes it rich in data on hazards, risks and controls. The advances in data mining and insight generation increasingly supported by artificial intelligence (AI) will enable more effective product safety management. The increasing use of AI with predictive analytics provides the prospect for enhanced hazard identification and targeted risk management that, if applied with sound judgement, will strengthen our ability to deliver safer products to the consumer.
The last 30 years has demonstrated that, despite the emergence of the occasional new hazard, the challenges we face remain remarkably consistent. Although we will undoubtedly benefit from technological advancements, we will inevitably continue to rely heavily on people to take the right actions and to make the right decisions to deliver safe products. I hope this review has provided some stimulus for continuous development in your systems, processes and, most importantly, people.
 |
AuthorAlec Kyriakides Independent Food Safety Consultant |